1. Development Background of Lithium-Ion Batteries
Emerging in the early 1970s, lithium-ion batteries revolutionized energy storage. Characterized by high energy density, extended cycle life, and long service duration, they power diverse applications: portable electronics, electric vehicles (EVs), smart wearables, and 3C products.
China’s “Carbon Neutrality” pledge (September 22, 2020) accelerated policy-driven decarbonization efforts. By 2022:
- Lithium-ion batteries dominated China’s new energy storage installations (93.7% market share)
- Accounted for 94% of newly deployed storage capacity
EV Market Expansion (2022):
- Total vehicles in operation: 319 million
- New energy vehicles (NEVs): 13.1 million (+67.13% YoY, 4.10% penetration)
- Battery electric vehicles (BEVs): 10.45 million (79.78% of NEVs)
The Dual Carbon Policy fuels massive investment and technological innovation across the lithium-ion battery sector, driving demand for core EV components.
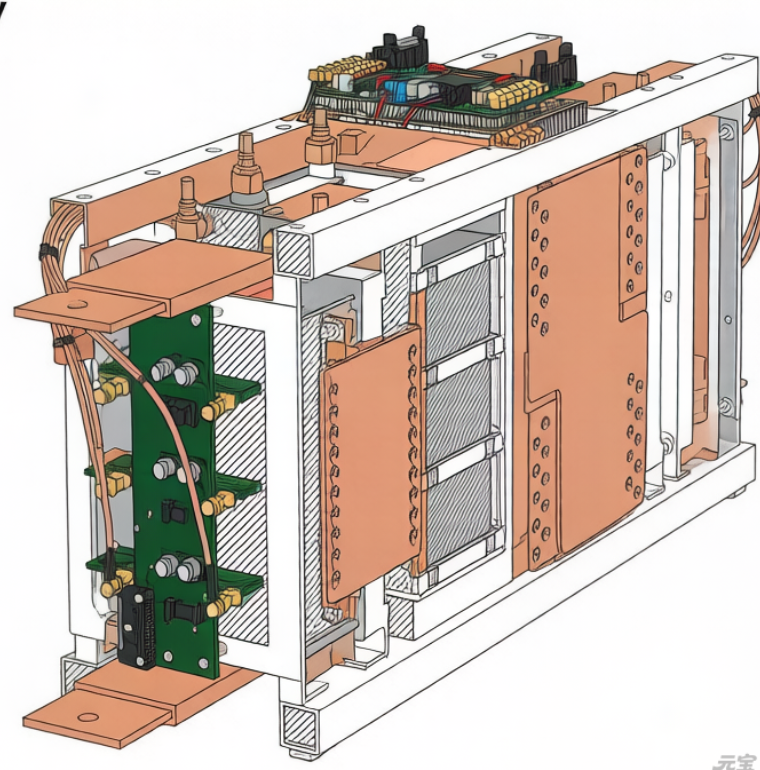
2.Structural Analysis of Lithium-Ion Batteries
In practical applications, lithium batteries are categorized by packaging into three types:
- Cylindrical
- Prismatic
- Pouch cells
Key differences lie in casing materials and winding configurations:
- Cylindrical/prismatic: Metal casings
- Pouch: Aluminum-laminated film encapsulation
Core Components:
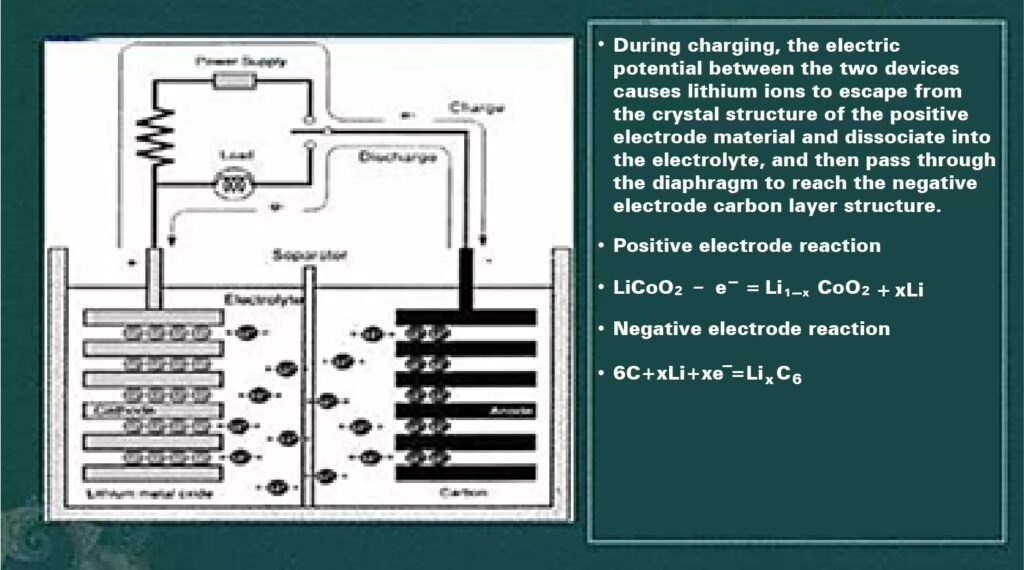
- Cathode
Materials: NMC, LiFePO₄, LiCoO₂, LiMn₂O₄
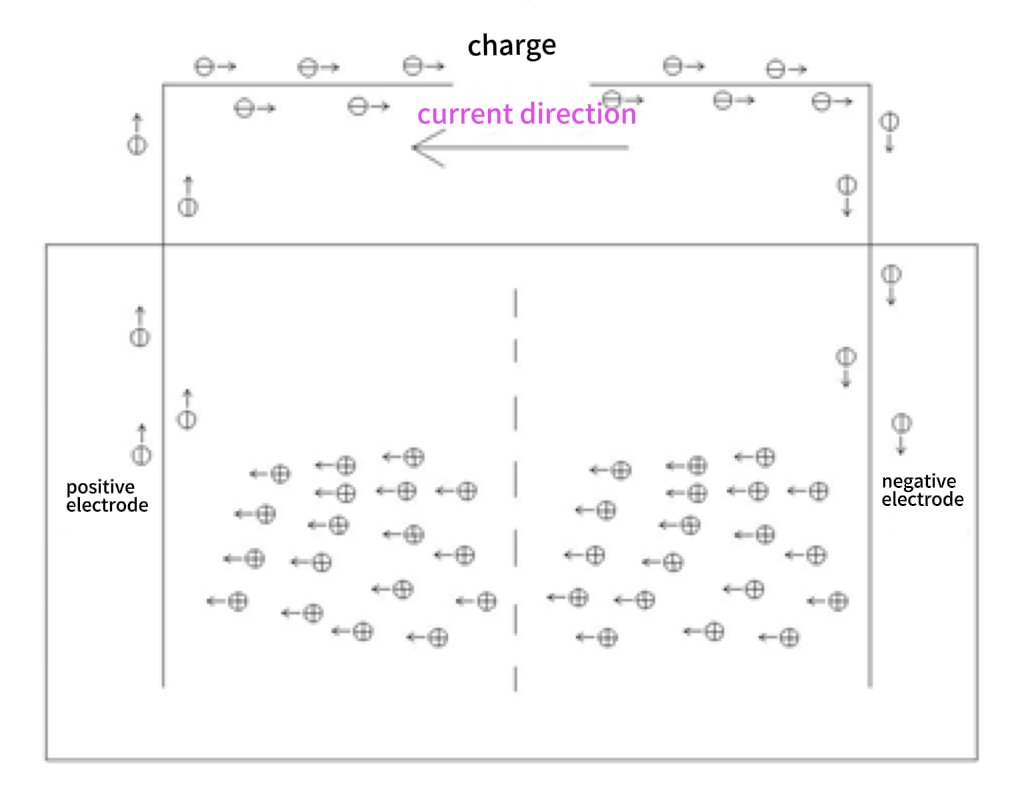
Function: During charging, lithium atoms dissociate into ions and electrons. Electrons return to the anode via external circuit, generating current. Reverse process occurs during discharge. - Anode
Material: Carbon-based substrates
Function: Electron storage/release - Separator
Materials: PP (polypropylene), PE (polyethylene), or PP/PE/PP composites
Function:- Prevents electrode contact/short-circuiting
- Enables ion transport while blocking electrons
Performance impact: Interface structure, internal resistance, capacity, cycle life, and safety
- Electrolyte
Function:- Ion transport medium between electrodes
- Critical for energy/power density
- Permeable to ions only (blocks electrons)
- Casing
Structural integrity and environmental protection
Battery operating principle diagram below:
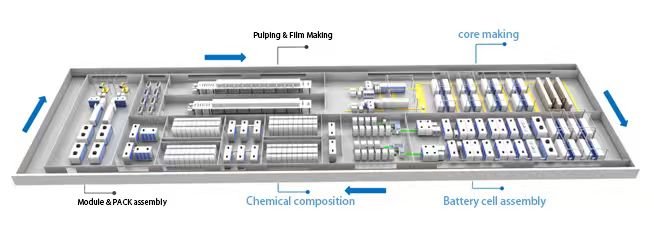
3.Lithium-Ion Battery Production
The most critical stages in lithium battery manufacturing are electrode production and cell assembly processes.
Key Processes:
Stacking:
Accommodates rigid electrode designs
Electrode Fabrication
Electrodes consist of conductive substrates coated with active material slurries.
Batteries convert chemical energy into electrical energy via electrochemical reactions.
Conductive materials are required to transfer this energy, with copper foil (anode) and aluminum foil (cathode) being optimal due to:
Superior electrical conductivity among metals
Cost-effectiveness
High ductility (essential for processing)
Cell Assembly Techniques
Winding:
Requires flexible electrodes to prevent brittle fracture during coiling
Lithium-Ion Battery Production Flow Diagram
Slurry Mixing (First Process)
The initial step involves mixing cathode/anode active materials, binders, and conductive additives into homogeneous slurries. Precise control of mixing duration and methodology is critical to ensure material uniformity. This process directly determines final battery quality and production yield.
Coating (Second Process)
Uniformly coat the mixed slurries onto aluminum foil (cathode) and copper foil (anode). Material selection rationale:
Copper foil provides stability at low potentials (anode)
Process control must guarantee consistent electrode thickness and coating weight to prevent cell performance variations.
Aluminum foil resists oxidation at high voltages (cathode)

Coating Process Schematic
Calendering(Third Process)
The formed electrodes undergo compression in a calender. This process is critical for enhancing energy density.

Calendering Process Schematic
Slitting(Forth Process)
Electrode sheets are cut to specified battery dimensions. This process generates microscopic burrs observable only under magnification. To prevent burrs from piercing separators—a critical safety hazard—burr formation must be strictly controlled. This concludes electrode manufacturing.
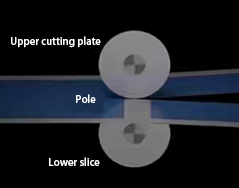
Slitting Process Schematic
Cell Assembly Process
Two primary methods are employed:
- Winding Method
- Layers stacked in sequence: Cathode → Separator → Anode → Separator
- Forms unassembled cells (jelly-roll)
- Requires CCD vision inspection systems for auto-alignment to prevent electrode misalignment
- Stacking Method
- Layers stacked: Cathode → Separator → Anode
Method Application Scope:
Stacking: Pouch cells
Winding: Cylindrical/prismatic cells

Winding Process Schematic
The unassembled cell is then placed into its casing, undergoes degassing, and is finally sealed after electrolyte injection and top cover placement.
4.Formation and Testing of Lithium-Ion Batteries
Following initial processing, the cell’s functional structure is complete. Subsequent stages include:
- Formation and capacity grading
- Performance testing
- Cell sorting
- Final assembly
These processes yield finished lithium-ion batteries with ensured safety and stable performance.
Formation Process Schematic
Formation Process
Following high-temperature aging, initial charging activates the battery, forming an SEI film on the anode surface. This electrolyte-derived layer:
- Blocks electrons and solvent molecules
- Permits only lithium-ion transport
Standard Procedure:
- Resting: 5 min on needle-bed formation fixture
- CC Charge: 0.02C to 3.0V
- Resting: 5 min
- CC Charge: Stepwise 0.05C/0.1C/0.15C to 3.5V
- Resting: 5 min
- CC Discharge: 0.1C to 50% SOC
- Aging: 15 days at ambient temperature
Optimization Insight:
Low-current charging (0.02C) initiates stable SEI formation. Subsequent moderate currents (0.05C–0.15C) enhance:
- Film density
- Thermal stability
- Ionic selectivity
Capacity Grading
Batteries undergo charge-discharge cycles using test equipment. Capacity is calculated via:
Capacity (Ah) = Discharge Current (A) × Cycle Duration (h)
Acceptance criteria:
- Measured capacity ≥ Design capacity
- Voltage/IR consistency
Final Validation
Post-formation batteries undergo:
- OCV (Open Circuit Voltage) testing
- DCIR (Direct Current Internal Resistance) measurement
- Case insulation verification
Qualified cells proceed to high-temperature aging, completing production.
5.Current Monitoring Solutions for Formation and Capacity Grading
Current detection technology is critical in formation and grading processes. Industry-standard precision for production lines is ±0.05%, achieved through two primary methods:
1. Sensor-Based Solution
- Employs high-precision closed-loop fluxgate sensors
- Typical accuracy: ±0.1–0.17% (insufficient standalone)
- Calibration enhancements:
- Piecewise linearity compensation
- Temperature curve-based correction
- Advantages: Compact size, stability, cost-effectiveness
- Limitation: PPM-grade sensors remain cost-prohibitive for mass deployment
2. Shunt-Based Solution
- Higher inherent accuracy
- Drawbacks:
- Significant thermal drift
- High power dissipation
- Primarily used in low-current scenarios or forced-cooling environments
Magtron’s Innovation
Proprietary iFluxgate® technology delivers:
- Cost-effective high-precision sensors
- Near-zero drift (±3ppm/°C typical)
- Wide bandwidth (DC-500kHz)
- Integrated SoC solutions for real-time industrial demands
Applications: EV, BESS, and industrial current/leakage monitoring with laboratory-grade accuracy.

References:
[1] Mao, S. K. Production Processes and Development Prospects of Lithium-Ion Batteries.
[2] Ye, J. X.; Qu, T.; Zhao, Y. F.; Shen, Z. A.; Li, Y. Y.; Ma, Q. H.; Wang, J. J. Study on High/Low-Temperature Performance of LiFePO₄ Cathode Material in Lithium-Ion Batteries.
[3] Wu, Z. Y.; Cao, S. X.; Wang, S. P.; Liu, Y.; Luo, G. Y.; Wu, Y. S.; Hu, X. D.; He, X. Y.; Shi, J. J.; Jiang, Y. M.; Liu, C. Development of Anode Materials for Fast-Charging Lithium-Ion Batteries.
[4] Liu, L.; Wang, H. L.; Zhang, Z. G. Working Principles and Key Materials of Lithium-Ion Batteries.
[5] Dai, Y. S.; Xian, Q. Y.; Huang, Z. M. Research on Performance of Lithium-Ion Batteries.
[6] Hu, G. X.; Xie, J. Y. Factors Influencing Safety of Lithium-Ion Batteries.
[7] Du, C. W.; Zhao, Y. J.; Wu, Y. S.; Liu, Q. G. Improved Electrochemical Performance of Natural Graphite via Heat Treatment.
[8] Huang, F.; Zhou, Y. H. Current Status and Developments in Electrolytes for Lithium-Ion Batteries.